Learning Management & Talent Development Insights
For 20 years, eLeaP® has helped organizations achieve excellence through learning, performance, & quality.

-
The 3 Q’s in Computer System Validation: A Comprehensive Guide to IQ, OQ, and PQ
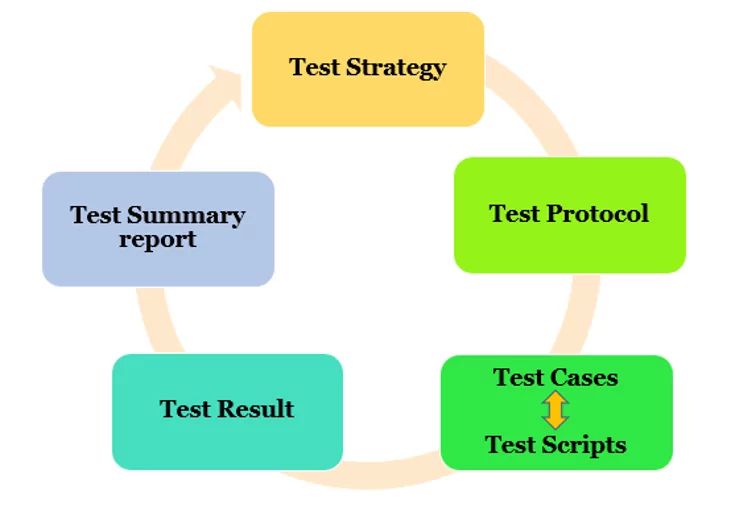
Executive Summary Computer System Validation (CSV) represents one of the most critical regulatory requirements facing FDA-regulated industries today. In an increasingly digital landscape where pharmaceutical manufacturing, clinical trials, and medical device development rely heavily on computerized systems, the stakes for proper validation have never been higher. At the heart of CSV lies the qualification process—specifically […]
-
9 Tips To Comply with 21 CFR Part 11

For highly regulated medical device organizations and life science industries, it has become a challenge to achieve 21 CFR Part 11 compliance and a matter of concern for companies subject to FDA inspections. Organizations must follow best practices in maintaining compliance and the quality process by following the 21 CFR Part 11 regulation. However, as […]
-
FDA Transition to Computer System Assurance: The New CSV

The 2003 FDA guidance paved the road for the implementation of efficient computer system validation for the pharmaceutical industry. Following that FDA guideline, the life science industry has become more efficient in validating computer systems and documentation in order to comply with regulations, inspections, compliance, and audits. The FDA launched the Case for Quality in […]
-
What Is a Validated LMS?

You may have heard the term “validated LMS” being thrown around when it comes to various types of platforms. This is an industry-specific term that’s applied to organizations with certain requirements for compliance and other protocols. Does your organization manufacture food, drugs, medical devices, or anything chemistry-related? You might want to pay close attention to […]
-
Achieving Compliance with 21 CFR Part 11

As the world is moving towards digitalization at a faster pace, the use of the computerized system in the life science industry brought a new perspective in terms of using a software application. Going digital has changed the way data is stored and managed. The transition to paperless recording has had a huge impact on […]