The 3 Q’s in Computer System Validation: A Comprehensive Guide to IQ, OQ, and PQ
Computer System Validation: Executive Summary
Computer System Validation (CSV) represents one of the most critical regulatory requirements facing FDA-regulated industries today. In an increasingly digital landscape where pharmaceutical manufacturing, clinical trials, and medical device development rely heavily on computerized systems, the stakes for proper validation have never been higher. At the heart of CSV lies the qualification process—specifically Installation Qualification (IQ), Operational Qualification (OQ), and Performance Qualification (PQ). These three sequential phases, collectively known as the “3 Q’s,” provide documented evidence that systems consistently perform their intended functions while maintaining the highest standards of data integrity and patient safety.
The qualification process is far more than a regulatory checkbox exercise. It represents a systematic approach to risk management that protects organizations from the devastating consequences of system failures, including FDA warning letters, consent decrees, product recalls, and, in the worst cases, harm to patients who depend on the safety and efficacy of regulated products. A single validation failure can cascade into millions of dollars in remediation costs, years of regulatory oversight, and irreparable damage to organizational reputation.
Key Takeaway: Successful IQ-OQ-PQ execution serves as your organization’s foundation for operational excellence, comprehensive risk mitigation, and sustained regulatory compliance across pharmaceutical, biotechnology, clinical research, and medical device industries. The investment in thorough qualification pays dividends through reduced operational risk, faster regulatory approvals, and the confidence that comes from knowing your systems will perform reliably when patient safety depends on them.
Take the course “The GAMP Approach to 21 CFR Part 11 Compliance” to stay current and relevant.
The Regulatory Landscape and Business Imperative
Understanding Computer System Validation in Context
Computer System Validation emerged from the recognition that computerized systems in regulated industries require the same level of control and verification as traditional manufacturing processes. The FDA’s 21 CFR Part 11 regulation, enacted in 1997 and refined through subsequent guidance documents, established that electronic records and signatures must be trustworthy, reliable, and generally equivalent to paper records and handwritten signatures.
This regulatory framework extends far beyond simple compliance requirements. It reflects a fundamental understanding that in industries where product quality directly impacts human health, every component of the manufacturing and development process—including the computerized systems that control, monitor, and document these processes—must demonstrate consistent, predictable performance.
The business case for comprehensive CSV extends well beyond regulatory compliance. Organizations with mature validation programs report significantly lower rates of system-related deviations, faster resolution of regulatory inspections, and reduced total cost of ownership for their technology investments. Conversely, companies with weak validation practices face not only regulatory consequences but also operational inefficiencies, data integrity questions, and the constant threat of system failures that can disrupt critical business processes.
The Evolution of Validation Thinking
The validation landscape has evolved considerably since the early days of 21 CFR Part 11. Initial approaches often applied one-size-fits-all methodologies that resulted in over-validation of low-risk systems and sometimes under-validation of truly critical applications. Modern validation thinking, exemplified by the FDA’s Computer System Assurance (CSA) initiative, emphasizes risk-proportionality and practical compliance strategies.
This evolution recognizes that validation resources—both time and expertise—are finite and should be applied where they provide the greatest risk reduction and business value. The contemporary approach to IQ-OQ-PQ reflects this thinking, scaling qualification activities based on system complexity, novelty, business criticality, and potential impact on product quality or patient safety.
Understanding Computer System Validation: Foundations and Framework
Defining Computer System Validation
Computer System Validation is a documented process that provides a high degree of assurance that a computerized system will consistently produce results meeting predetermined specifications and quality attributes. This definition, while seemingly straightforward, encompasses a complex web of activities, documentation, and verification procedures that span the entire system lifecycle from initial requirements through retirement.
The validation process operates on several fundamental principles that distinguish it from standard software testing or quality assurance activities. First, validation emphasizes documented evidence over informal verification. Every aspect of system performance must be formally tested, recorded, and approved by qualified personnel. Second, validation follows a risk-based approach that recognizes not all systems pose equal risks to product quality or patient safety. Third, validation creates traceability from high-level business requirements through detailed technical specifications to specific test cases and results.
The Risk-Based Validation Paradigm
Modern CSV practice has moved decisively toward risk-based approaches that recognize the fundamental differences between various types of computerized systems. This paradigm shift acknowledges that a simple spreadsheet used for non-GxP calculations requires vastly different validation attention than a manufacturing execution system controlling critical production processes.
Risk assessment in CSV considers multiple factors, including system complexity, degree of customization, novelty of the technology, supplier assessment results, and potential impact on product quality or patient safety. Systems are typically categorized into risk levels that determine the appropriate validation approach:
Category 1 – Infrastructure Software: Operating systems, database management systems, and other foundational software components typically require minimal validation beyond verification of proper installation and configuration.
Category 2 – Standard Commercial Software: Off-the-shelf applications with minimal configuration require moderate validation focusing on configuration verification and functional testing of configured features.
Category 3 – Configured Commercial Software: Standard applications with significant configuration or customization require comprehensive validation including extensive functional testing and configuration verification.
Category 4 – Custom Software: Bespoke applications developed specifically for the organization require the most comprehensive validation including code reviews, extensive testing, and detailed documentation.
Category 5 – Embedded Software: Software embedded in instruments or equipment requires specialized validation approaches that may include supplier assessment and verification of embedded software quality systems.
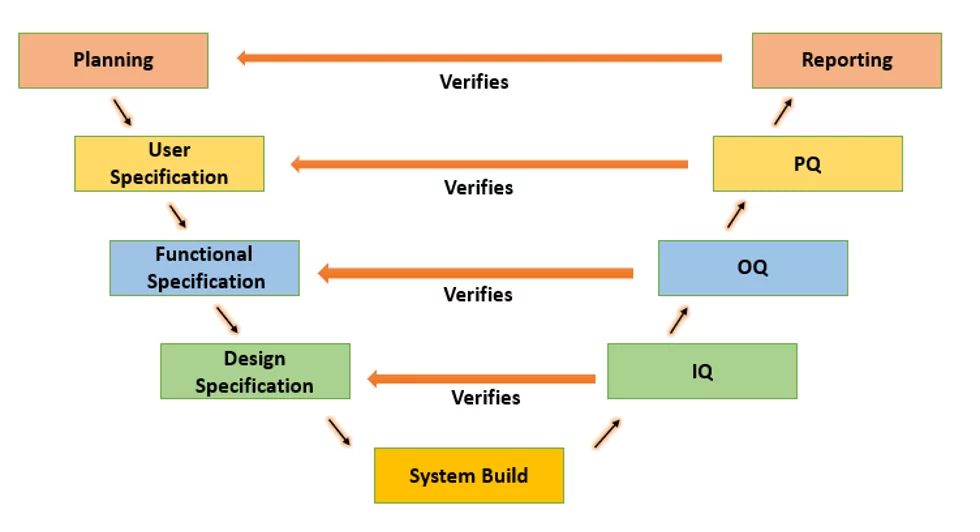
Visual Overview of IQ-OQ-PQ Activities
Understanding the flow and relationships between the three qualification phases is crucial for effective validation planning. The diagram below provides a clear visual representation of what each phase accomplishes, who is involved, and when each activity occurs in the validation sequence.
This visual representation clearly illustrates several key concepts:
Sequential Dependency: Each phase builds upon the previous one, with IQ establishing the foundation for OQ testing, and OQ providing the functional verification necessary for meaningful PQ validation.
Stakeholder Involvement: Different stakeholders play primary roles in each phase, from technical teams during IQ to end users during PQ, ensuring comprehensive validation coverage.
Environment Progression: Testing environments progress from development/test settings during IQ to validation environments for OQ, culminating in production or production-like environments for PQ.
Documentation Requirements: Each phase generates specific deliverables that support subsequent phases and provide the documented evidence required for regulatory compliance.
Objectives of Performing 3 Q’s
Performing the IQ-OQ-PQ is an integral part of testing activities. Testing is often performed at several levels depending on the risk, complexity, and novelty. One level is appropriate for simple and low-risk systems, while multiple levels may be required for complex, configured, or custom systems. Testing must be carried out in accordance with the test strategy documented in the computer system validation plan.
The primary objectives include:
- Identifying defects so they can be corrected or removed before operational use
- Preventing failures that might affect patient safety, product quality, or data integrity
- Providing documented evidence that the system performs as specified
- Demonstrating that the system meets its requirements
- Providing confidence that the system is fit for its intended use
- Providing a basis for user acceptance
- Meeting a critical regulatory requirement
What are IQ, OQ, and PQ?
IQ, OQ, and PQ are sequential activities that are carried out to validate the system. IQ stands for Installation Qualification, OQ for Operational Qualification, and PQ for Performance Qualification.
Deep Dive: The Three Qualification Phases
Installation Qualification (IQ): Building the Foundation
Installation Qualification represents the first critical phase in the qualification process, establishing the foundation upon which all subsequent validation activities build. The primary objective of IQ is to verify and document that system components are correctly installed, properly configured, and ready for functional testing.
Comprehensive Installation Verification
The scope of Installation Qualification extends far beyond simple software installation. Modern computerized systems typically involve complex architectures including multiple servers, database systems, network components, security infrastructure, and integration points with existing systems. Each of these components must be verified against approved specifications and documented to create a comprehensive baseline configuration.
Hardware verification includes confirming that servers meet minimum specifications for processing power, memory, storage capacity, and network connectivity. This verification must extend to redundancy and backup systems, ensuring that disaster recovery capabilities are properly installed and configured. Environmental factors such as power conditioning, HVAC requirements, and physical security measures must also be verified and documented.
Software installation verification encompasses not only the primary application but also all supporting software, including operating systems, database management systems, middleware, and integration components. Version control is critical, with documentation required for all software versions, patches, and configuration settings. This documentation serves as the baseline for ongoing configuration management and change control processes.
Detailed IQ Test Examples
Hardware Verification Tests:
- Verify database server configuration: Minimum 64GB RAM, 2TB SSD storage, dual network interfaces
- Confirm backup server specifications: Redundant power supplies, RAID 6 disk configuration, 72-hour battery backup
- Test network connectivity: Verify 1Gbps network speed, latency under 5ms to critical systems
- Validate environmental controls: Temperature maintained at 68-72°F, humidity 45-55%, clean power with UPS backup
Software Installation Tests:
- Confirm application version 3.2.1 with security patches SP-7, SP-8, and SP-9 applied
- Verify database version PostgreSQL 13.4 with proper table spaces and user permissions configured
- Test integration middleware: Message queuing service running with proper failover configuration
- Validate antivirus software: Real-time scanning enabled, definition updates automated, exclusions properly configured
Security Configuration Tests:
- Verify user account setup: Administrator, power user, and standard user roles are properly configured
- Test password policy enforcement: Minimum 12 characters, complexity requirements, 90-day expiration
- Confirm audit trail functionality: All user actions logged with timestamps, user identification, and data changes recorded
- Validate encryption settings: Data encrypted at rest using AES-256, TLS 1.3 for data in transit
Installation testing should verify that the following documents are available where appropriate:
- User and technical guides
- Standard operating procedures
- Training schedules
- Service level agreements
- Security procedures
- Hardware inventory
- Specification sheets
- Program source code
- Backup and restore procedures
- Data archival and retrieval capabilities
- Ability to deal with high-volume load
Operational Qualification (OQ): Proving Functionality
Operational Qualification represents the most comprehensive phase of the qualification process, systematically verifying that all system functionality performs correctly according to approved specifications. This phase employs rigorous testing methodologies to ensure that systems can reliably perform all required functions under both normal and exceptional conditions.
Operational Qualification testing is also referred to as Functional Testing. OQ tests confirm that all functionality defined in the Functional Specification is working correctly and that there are no bugs. The functionality tested should support the specific business process, and based on the risk and supplier assessment, the level of testing is defined. Generally, black box testing is performed for functional specification testing.
As suggested by the FDA, specific types of testing should be performed depending on risk, complexity, novelty, and supplier assessment of the system:
- Normal case testing (Positive or Capability testing)
- Invalid case testing (Negative or Resistance testing)
- Repeatability testing
- Performance testing
- Volume/load testing
- Regression testing
- Structural/path testing
Advanced Testing Methodologies
Boundary Value Testing: This methodology specifically tests system behavior at the boundaries of acceptable input ranges. For example, if a system accepts temperatures between 2°C and 8°C, boundary value testing would test 1.9°C, 2.0°C, 2.1°C, 7.9°C, 8.0°C, and 8.1°C to ensure proper handling of edge cases.
State Transition Testing: Complex systems often maintain internal states that affect their behavior. State transition testing systematically verifies that systems transition correctly between operational states and that state-dependent functionality operates correctly in each state.
Error Recovery Testing: Systems must handle error conditions gracefully without compromising data integrity or system stability. Error recovery testing intentionally introduces various error conditions to verify that systems respond appropriately and can recover to normal operation.
Detailed OQ Testing Examples
Normal Case Testing (Positive Testing): User Management Functions:
- Create new user accounts with valid information: Username “jsmith”, email “john.smith@company.com”, role “Analyst”
- Successful login with valid credentials returns the appropriate dashboard and menu options
- Password reset functionality sends a secure reset link to the registered email address
Data Entry and Processing:
- Laboratory results entry: pH value 7.2, temperature 25.3°C, pressure 1.013 bar, saved correctly with automatic timestamp
- Batch record creation: Lot number LOT2024001, product code PRD-ASP-500, quantity 10,000 units generates proper batch documentation
- Electronic signature application: User “mwilson” signs batch record with password authentication, creates tamper-evident signature record
Invalid Case Testing (Negative Testing): Security and Access Control:
- Invalid login attempts: Username “hacker” with password “password123” displays a generic “Invalid credentials” message
- Account lockout: Five consecutive failed login attempts lock the account for 30 minutes and notify the security administrator
- Unauthorized access attempts: User with “Analyst” role attempting to access “Administrator” functions receives an ” access denied message
Data Validation and Integrity:
- Invalid data entry: pH value “-5.0” displays error message “pH must be between 0 and 14” and prevents record save
- Required field validation: The temperature field left blank displays the “Temperature is required” error and highlights the field
- Data format validation: Date entry “February 30, 2024” displays “Invalid date” error and suggests correct format
Performance Qualification (PQ): Real-World Validation
Performance Qualification represents the final and most critical phase of the qualification process, demonstrating that systems perform effectively in their actual operating environment under realistic operational conditions. This phase bridges the gap between controlled testing environments and real-world operational use, providing the final verification that systems are truly ready for production deployment.
Performance Qualification testing is called User Acceptance testing. It confirms that the software will meet the user’s needs and is fit for their intended use, as defined in the User Requirements Specification.
Factory Acceptance Testing (FAT) and Site Acceptance Testing (SAT)
The acceptance testing may be carried out in two stages:
Factory Acceptance Testing is performed at the supplier’s site before delivery to demonstrate that the system is sufficiently operational to be installed and tested on-site. This testing provides early verification that core system functionality meets requirements and can identify major issues before the expense and complexity of site installation.
Site Acceptance Testing, also called System Acceptance Testing, demonstrates that the system is functioning in its operational environment and interfaces correctly with other systems. SAT testing scenarios must reflect actual business processes and operational conditions as closely as possible while maintaining appropriate controls for testing activities.
Advanced PQ Testing Scenarios
Complete Business Process Execution: Manufacturing Batch Process:
- Execute a complete batch from raw material receipt through final product release
- Verify material tracking throughout the manufacturing process with proper genealogy records
- Test batch record generation, review, approval, and electronic signature application
- Confirm integration with the laboratory information management system for testing coordination
- Validate the final batch release process, including quality assurance review and approval
Peak Load and Stress Testing: Maximum Operational Conditions:
- Test system performance during peak usage periods with maximum concurrent users
- Verify database performance with maximum expected data volumes and query complexity
- Test backup and recovery procedures under operational load conditions
- Confirm system stability during extended continuous operation (72-hour stress test)
- Validate failover and redundancy systems under operational stress conditions
How to Perform IQ-OQ-PQ: FDA-Recommended Framework
According to FDA guidelines, the test strategy should be established with an appropriate approach for testing each requirement through the systematic application of IQ-OQ-PQ methodologies. The FDA has defined a typical structure for testing documentation that ensures comprehensive validation coverage while maintaining regulatory compliance.
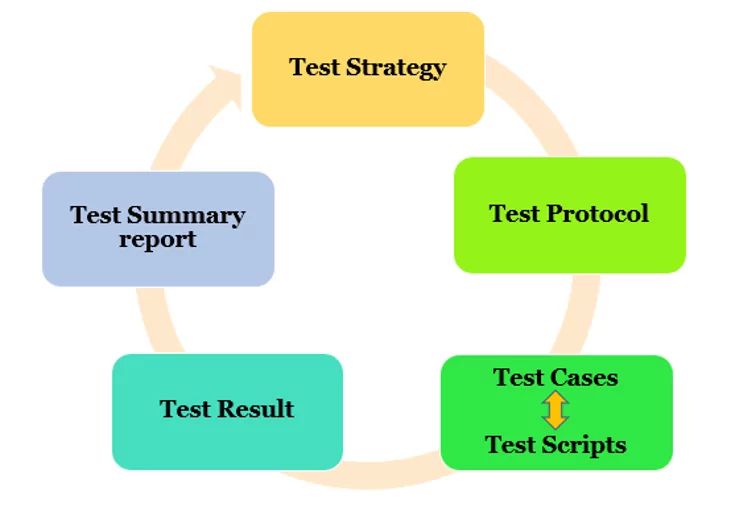
The verification of IQ-OQ-PQ is carried out through a systematic approach that follows this documented framework:
Core Documentation Framework
Test Strategy: The overarching approach that defines how validation will be conducted, including risk assessment results, testing scope, and resource allocation decisions.
Test Protocol: Detailed procedures that specify what will be tested, how testing will be conducted, acceptance criteria, and roles and responsibilities for execution.
Test Cases and Scripts: Specific step-by-step procedures that testers follow to verify system functionality, with clearly defined inputs, expected outputs, and pass/fail criteria.
Test Results: Documented evidence of test execution, including actual results, deviations from expected results, and resolution of any issues identified during testing.
Test Summary Report: Comprehensive analysis of all testing activities with conclusions about system readiness for operational use and any recommendations for ongoing monitoring or maintenance.
Protocol Requirements
Installation Qualification Protocol: The Installation Qualification Protocol verifies the proper installation and configuration of a System. Depending on the system’s needs and complexity, Installation Qualification can be combined with Operational Qualification or Performance Qualification. Installation Qualification protocols should be approved before protocol execution. A copy of the unexecuted protocol should be kept in the validation package. The System Owner and Quality Assurance should approve the unexecuted protocol. The executed protocol should be signed by the tester and reviewed by the system owner and Quality. Installation Qualification must be performed before completing the Operational Qualification or Performance Qualification.
Operational Qualification Protocol: The Operational Qualification Protocol is a collection of test cases used to verify the system’s functionality. The operational qualification test requirements are defined in the Functional Requirements Specification. Operational Qualification is usually performed before the system is released for use.
Performance Qualification Protocol: Performance Qualifications are a collection of test cases that verify that a system performs as expected under simulated real-world conditions. The performance qualification test requirements are defined in the User Requirements Specification (or possibly the Functional Requirements Specification).
The GAMP V-Model Framework: Ensuring Comprehensive Validation
Understanding the V-Model Architecture
The GAMP V-Model provides a systematic framework for organizing validation activities that ensures comprehensive coverage of all system requirements while maintaining traceability between requirements and testing. This model has become the industry standard for structuring validation projects because it provides clear guidance on what should be tested and when, while ensuring that every requirement has corresponding verification.
Process validation aims to establish documented evidence that the system meets a set of defined requirements. The defined discipline for a validation process has proven to be the ideal way to guarantee the best quality of the validated system. Each phase of the validation process includes documentation with detailed results of each qualification test. To understand the qualification process, we should know more about the testing strategy defined by the FDA. The classic “V Diagram” was popularized by industry organizations, such as ISPE, through GAMP Guides. This “V Diagram” explains which requirements are tested against which types of requirements. The V-Model derives its name from its visual representation, which resembles the letter “V” in a diagram. The left side of the V represents the requirements development process, starting with high-level user requirements and progressing through increasingly detailed specifications. The right side of the V represents the testing activities, with each level of requirements corresponding to a level of testing.
Take the course “The GAMP Approach to 21 CFR Part 11 Compliance” to stay current and relevant.
Requirements Traceability Matrix
The foundation of effective V-Model implementation lies in comprehensive requirements traceability. Each requirement must be clearly defined, uniquely identified, and linked to corresponding test cases that verify its implementation.
User Requirements Specification (URS) Traceability: The URS defines what the system must do from the user’s perspective, focusing on business processes and operational needs. Each URS requirement must be traced to corresponding Performance Qualification test cases that demonstrate the system meets user needs in operational environments.
Functional Requirements Specification (FRS) Traceability: The FRS defines how the system will implement user requirements, specifying detailed system behavior and functionality. Each FRS requirement must be traced to corresponding Operational Qualification test cases that verify functional implementation.
Design Specification Traceability: Design specifications define technical implementation details, including database schemas, user interface designs, integration specifications, and system architecture. Each design specification element must be traced to the corresponding Installation Qualification verification activities.
Comparison: IQ vs OQ vs PQ
| Aspect | Installation Qualification (IQ) | Operational Qualification (OQ) | Performance Qualification (PQ) |
| Primary Focus | System installation and configuration | Feature functionality and behavior | Real-world performance and acceptance |
| When Performed | After installation, before OQ | After IQ completion | After OQ completion |
| Testing Environment | Validation/test environment | Controlled test environment | Production or production-like environment |
| Who Executes | Installation team and validators | Functional testers and validators | End users and validators |
| Key Deliverables | Installation documentation, configuration baseline | Functional test results, defect reports | User acceptance evidence, performance metrics |
| Typical Duration | 1-2 weeks | 2-6 weeks (varies by complexity) | 1-4 weeks |
Risk-Based Qualification Strategies
Evolution of Risk-Based Thinking
The pharmaceutical and medical device industries have undergone a fundamental shift in validation thinking over the past two decades, moving from prescriptive, one-size-fits-all approaches to risk-based strategies that scale validation efforts according to actual risk and business impact.
Traditional validation approaches often applied identical methodologies to all computerized systems regardless of their complexity, business criticality, or potential impact on product quality or patient safety. This approach resulted in overvalidation of simple, low-risk systems and, at times, undervalidation of complex, high-risk applications.
Risk-based validation recognizes that different systems present different levels of risk and require correspondingly different levels of validation attention. A simple spreadsheet used for non-GxP calculations requires minimal validation compared to a manufacturing execution system that controls critical production processes.
Implementation Strategies for Different Risk Categories
Low-Risk System Validation (Category 1-2 Systems):
- Streamlined IQ approach with supplier-provided installation checklists
- Focused OQ testing on configured functionality and critical business processes
- Simplified PQ verification with user acceptance testing for key business processes
Medium-Risk System Validation (Category 3 Systems):
- Comprehensive IQ verification with detailed hardware and software installation verification
- Thorough OQ testing, including comprehensive functional testing and security assessment
- Complete PQ validation with end-to-end business process testing and realistic data volumes
High-Risk System Validation (Category 4-5 Systems):
- Extensive IQ verification, including detailed verification of all components and source code review
- Comprehensive OQ testing with complete functional testing and extensive security testing
- Rigorous PQ validation with complete end-to-end process validation and extended stability testing
Common Challenges and Practical Solutions
Challenge 1: Resource Constraints and Timeline Pressure
Problem: Organizations often underestimate the time and resources required for comprehensive qualification, leading to rushed testing that may miss critical issues.
Solutions:
- Develop comprehensive validation effort estimation models based on historical project data
- Implement standardized validation processes and documentation templates
- Invest in validation management tools that automate routine tasks
- Focus validation resources on the highest-risk system components and functions
Challenge 2: Vendor Coordination and Documentation Gaps
Problem: Inadequate vendor documentation or support during qualification phases.
Solutions:
- Establish clear validation support requirements in vendor contracts
- Develop standardized vendor assessment procedures
- Implement comprehensive vendor audit programs
- Require vendors to provide qualified technical support during validation activities
Challenge 3: Change Management During Qualification
Problem: System changes occurring during validation can invalidate completed testing.
Solutions:
- Establish formal change control processes with impact assessment procedures
- Implement change classification systems based on potential validation impact
- Develop streamlined revalidation procedures for common types of changes
- Create change tracking systems for comprehensive documentation
Modern Considerations: Computer System Assurance (CSA)
The Evolution of Computer System Assurance
The FDA’s Computer System Assurance initiative represents a fundamental shift in regulatory thinking about computerized system validation, emphasizing practical, risk-based approaches that focus on actual risk mitigation rather than procedural compliance.
Core CSA Principles:
- Risk-Proportionate Approach: Scaling validation activities based on actual risk rather than applying uniform approaches to all systems
- Leveraging Supplier Quality Systems: Utilizing supplier testing and documentation to reduce customer validation requirements
- Continuous Monitoring and Lifecycle Management: Emphasizing ongoing system monitoring rather than point-in-time validation
Emerging Technologies and Validation Challenges
Cloud Computing and SaaS: Present unique validation challenges, including limited customer control over infrastructure and shared responsibility models.
Artificial Intelligence and Machine Learning: Require validation approaches that address algorithm transparency, training data validation, and ongoing performance monitoring.
Internet of Things (IoT) and Edge Computing: Present challenges include device management, network security, and data integrity across distributed systems.
Implementation Roadmap
Phase 1: Strategic Planning and Preparation (4-8 weeks)
- Develop validation master plans
- Assemble cross-functional validation teams
- Select validation management tools and testing environments
Phase 2: Risk Assessment and Validation Planning (2-4 weeks)
- Conduct systematic risk assessments
- Define validation approaches for each system component
- Develop comprehensive protocol plans
Phase 3: Installation Qualification Execution (1-4 weeks)
- Execute installation verification procedures systematically
- Document complete system configuration
- Establish a configuration baseline for change control
Phase 4: Operational Qualification Execution (3-8 weeks)
- Execute comprehensive functional testing
- Conduct integration and interface testing
- Address test failures and perform retesting
Phase 5: Performance Qualification Execution (2-6 weeks)
- Coordinate user acceptance testing
- Conduct operational readiness verification
- Complete final documentation and approvals
Phase 6: Documentation and Closure (1-2 weeks)
- Compile final validation package
- Obtain required approvals and signatures
- Transfer system to operational support
Best Practices for IQ-OQ-PQ Execution
- Tests should be executed according to pre-defined and pre-approved specifications
- Each test should be run according to the test script, and all test results should be recorded
- Tests should be performed by trained testers with appropriate qualifications
- Test results must be documented as and when testing occurs, and must be retained
- All test results should be immediately and accurately recorded in an indelible format
- In case of any corrections, they should be crossed out with a single line, signed, and dated with a brief justification
- The script should clearly state if the test is passed or failed with no ambiguity
- In case of a failed test, a defect must be raised and tracked through final closure with retesting
- A summary report should be produced for each qualification test performed, stating all findings and conclusions
- The executed protocol and reports must be signed and approved by the Quality representative
Significance of IQ-OQ-PQ
IQ-OQ-PQ requires strict adherence to the process along with proper documentation. While the basics of IQ-OQ-PQ are critical to understand and implement, it is also important to acknowledge the challenges encountered in performing these activities. As industries are required to follow FDA guidance, product quality and performance, delivery precision, and patient safety are of the utmost importance.
Quality assurance professionals must ensure that validation activities are planned, executed, and documented according to established standards. Even if the product or software has passed all verification stages and fails to demonstrate compliance in one aspect of IQ-OQ-PQ, the result can be disastrous and incur considerable costs for the organization. Hence, the successful completion of IQ-OQ-PQ validation is fundamental to the successful delivery of the system from development to end users.
Conclusion and Strategic Recommendations
The landscape of computer system validation continues to evolve rapidly, driven by technological innovation, regulatory modernization, and increasing sophistication in risk-based approaches. Organizations that embrace this evolution while maintaining fundamental validation principles will gain significant competitive advantages through improved operational efficiency, reduced regulatory risk, and enhanced capability to adopt new technologies.
Immediate Action Priorities
- Assess and modernize current validation approaches to incorporate risk-based principles and contemporary best practices
- Invest systematically in validation capabilities, including personnel, processes, and technology infrastructure
- Build strategic partnerships with suppliers, consultants, and regulatory authorities that support validation excellence
- Embrace innovation while maintaining fundamental commitment to quality, compliance, and patient safety
- Develop an organizational culture that views validation as a strategic capability rather than a compliance burden
Success in computer system validation ultimately depends on recognizing that validation is not about checking regulatory boxes or meeting documentation requirements—it is about building operational excellence that supports the mission of delivering safe, effective products to patients who depend on them. The 3 Q’s of IQ-OQ-PQ provide the systematic framework for achieving this excellence, but success requires organizational commitment, appropriate investment, and sustained focus on continuous improvement.
Appendix A: Practical Implementation Templates
Risk Assessment Template
System Information:
- System Name: _______________
- Version: _______________
- Supplier: _______________
- Business Function: _______________
- Implementation Date: _______________
Risk Factors Assessment (Score 1-5, where 5 = highest risk):
System Complexity:
- Architecture complexity: ___
- Customization level: ___
- Integration complexity: ___
- Technology novelty: ___
Business Criticality:
- Patient safety impact: ___
- Product quality impact: ___
- Regulatory significance: ___
- Business continuity impact: ___
Supplier Assessment:
- Quality system maturity: ___
- Regulatory compliance history: ___
- Technical support capability: ___
- Validation documentation quality: ___
Overall Risk Score: _____ (Sum of all factors) Risk Category:
- Low Risk (20-35): Streamlined validation approach
- Medium Risk (36-55): Standard validation approach
- High Risk (56-80): Comprehensive validation approach
IQ Protocol Template
Installation Qualification Protocol System: _______________ Version: _______________ Protocol Version: _______________ Date: _______________
Objective: To verify that [System Name] has been installed correctly according to approved specifications and is ready for operational qualification testing.
Scope: This protocol covers verification of:
- Hardware installation and configuration
- Software installation and configuration
- Security configuration
- Integration setup
- Documentation completeness
OQ Protocol Template
Operational Qualification Protocol System: _______________ Version: _______________ Protocol Version: _______________ Date: _______________
Objective: To verify that [System Name] performs all required functions correctly according to functional specifications.
Test Categories:
- Functional Testing
- Performance Testing
- Security Testing
PQ Protocol Template
Performance Qualification Protocol System: _______________ Version: _______________ Protocol Version: _______________ Date: _______________
Objective: To demonstrate that [System Name] performs effectively in the operational environment and meets user requirements.
Appendix B: Regulatory Reference Guide
Key Regulatory Documents
FDA Guidance Documents:
- General Principles of Software Validation (2002)
- Computerized Systems Used in Clinical Investigations (2007)
- Data Integrity and Compliance with Drug CGMP (2018)</li>
- Com
- puter Software Assurance for Manufacturing and Quality Operations (2022)
International Guidelines:
- ICH Q7 Good Manufacturing Practice Guide (2000)
- ICH E6(R2) Good Clinical Practice (2016)
- EMA Questions and Answers on Computer System Validation (2010)
- PIC/S Recommendation on Computerised Systems (2007)
Industry Standards:
- ISPE GAMP 5: A Risk-Based Approach to Compliant GxP Computerized Systems (2008)
- IEEE 1012: Standard for System and Software Verification and Validation (2016)
- ISO 14155: Clinical Investigation of Medical Devices for Human Subjects (2020)
Glossary of Terms and Acronyms
21 CFR Part 11: FDA regulation establishing requirements for electronic records and electronic signatures in FDA-regulated industries.
<strong>Computer System Assurance (CSA): FDA’s modern, risk-based approach to computerized system validation, emphasizing practical compliance strategies.
<strong>Computer System Validation (CSV): Documented process providing assurance that computerized systems consistently perform their intended functions.
Installation Qualification (IQ): Verification that system components are installed correctly according to approved specifications.
Operational Qualification (OQ): Verification that the system functions correctly according to functional specifications under normal and abnormal conditions.
Performance Qualification (PQ): Demonstration that the system performs effectively in an operational environment under realistic conditions.
Risk-Based Validation: An approach that scales validation efforts based on system complexity, novelty, and potential impact on product quality or patient safety.