21 CFR Part 11 LMS
Power your Part 11 compliance with eLeaP


21 CFR Part 11 Compliant Learning Management System
Unlock the potential of seamless regulatory compliance and enhanced learning experiences with eLeaP’s 21 CFR Part 11-compliant Learning Management System (LMS). We have meticulously designed our platform to meet the stringent requirements set forth by the FDA, ensuring that your organization navigates the regulatory landscape effortlessly while fostering a culture of continuous learning and growth.
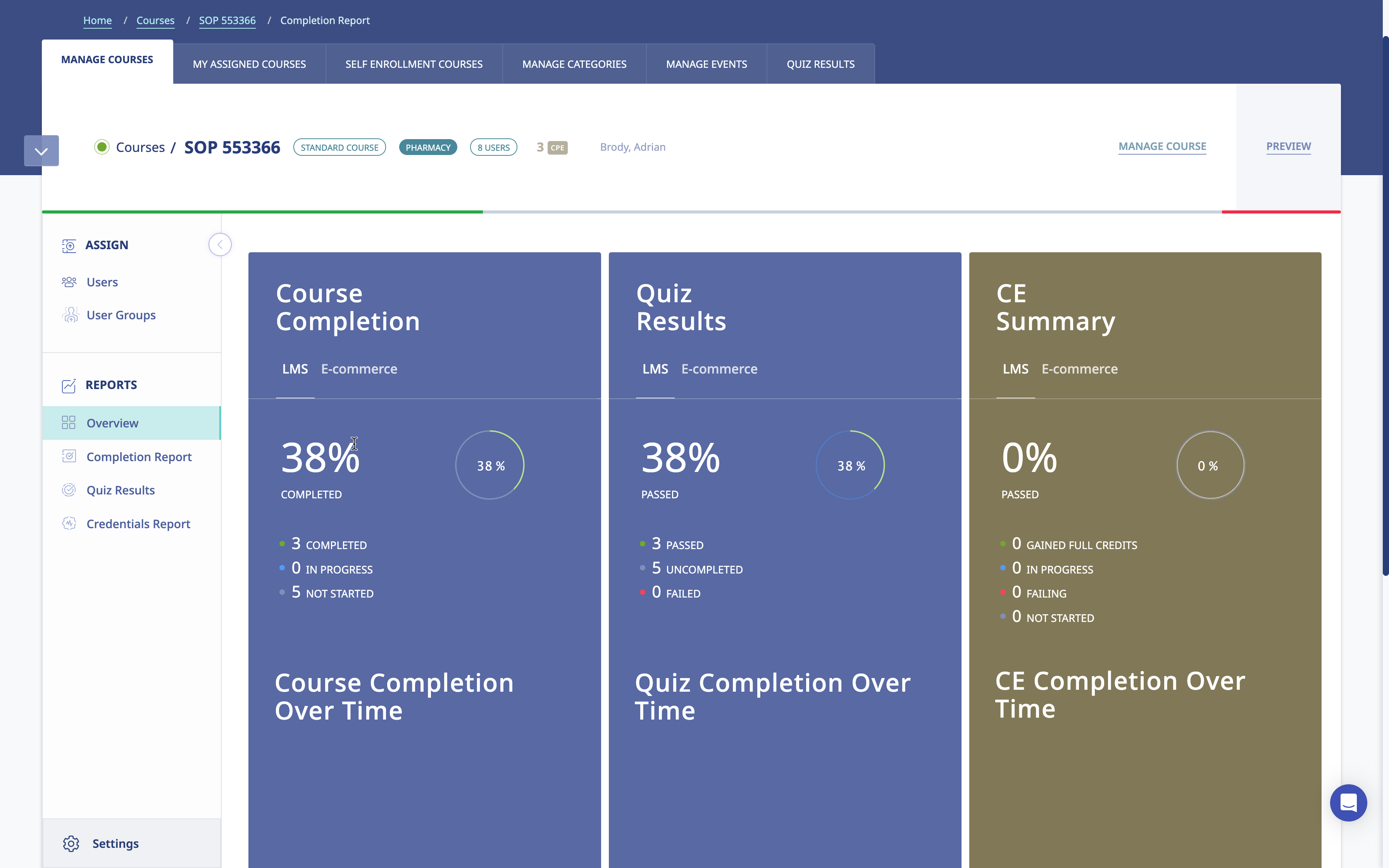
Key Features
-
Electronic Signatures
Ensure authenticity and accountability with secure electronic signatures, meeting 21 CFR regulatory requirements while maintaining traceability.
-
Robust Audit Trails
Track and record every action and access within the system, providing comprehensive audit trails for transparency and compliance.
-
Role-Based Access Control
Defined access levels based on roles, safeguarding data integrity and ensuring confidentiality across the platform.
-
Document Management
Organize and manage documents securely within the system, facilitating easy access, revisions and compliance with document control standards.
-
Training Records Management
Streamline record-keeping with centralized training records management, simplifying compliance reporting and audits.
eLeaP’s 21 CFR Part 11-compliant Learning Management System provides a comprehensive solution that harmonizes regulatory compliance with unparalleled learning experiences. Need GxP, Validation & Advisory services? For over 19 years, our platform has streamlined compliance efforts by integrating electronic signatures, robust audit trails, role-based access control, document management, and training record management.
Benefits of eLeaP’s 21 CFR Part 11 Learning Management System
Organizations seeking compliance and efficient learning management can benefit from the following advantages:
-
Cost-Efficiency and Time Savings
Streamline compliance processes and reduce manual efforts. Our LMS automates compliance tasks, saving time and resources while reducing the risk of errors.
-
Global Accessibility and Scalability
Enable learning from anywhere, anytime. With a cloud-based infrastructure, our platform ensures worldwide accessibility and scalability, accommodating the needs of your growing organization.
-
Enhanced Training Effectiveness
Elevate training effectiveness through interactive multimedia content, assessments, and real-time feedback, ensuring better knowledge retention and application.
-
Regulatory-Grade Security Measures
Rest assured of top-notch security measures. Our LMS employs encryption, intrusion detection, and regular security updates, automatic back-ups to keep your data protected.
-
Tailored Learning Paths for Diverse Audiences
Create personalized learning paths for different user groups, ensuring each learner receives relevant and targeted training.
Join many industry leaders who trust eLeaP to revolutionize compliance and learning management. Experience the synergy of regulatory compliance and enhanced learning outcomes with our 21 CFR Part 11-compliant LMS.
Platform Strengths
Discover the inherent advantages and standout qualities of the eLeaP 21 CFR Part 11 platform.
Organizations benefit from reduced compliance risks, significant time and cost savings through automation, enhanced data security, and personalized learning paths, ultimately ensuring regulatory adherence without compromising on the quality of learning experiences.
Life Science Industry
In the rapidly evolving life sciences sector, compliance and innovation are the pillars of success. For over 19 years, our 21 CFR Part 11-compliant learning management system has supported organizations in the industry. Here’s how:
-
Regulatory Compliance
Comply with mandates using electronic signatures, audit trails, and role-based access control, ensuring adherence to stringent regulatory requirements.
-
Data Integrity Fortification
Safeguard sensitive information with robust document management, ensuring secure storage and easy access to critical documents while maintaining data integrity.
-
Training Precision
Tailor learning experiences to suit diverse learner groups, enabling comprehensive training records management for compliance reporting and audits.
-
Streamlined Operations
Optimize workflows and reduce compliance risks with automated processes, enabling seamless integration and efficient management of learning resources.
-
Enhanced Security Protocols
Elevate data security through stringent access controls, encryption measures, and continuous updates, fortifying your platform against potential threats.
Experience a transformative shift in the life sciences landscape with eLeaP’s 21 CFR Part 11 LMS, where compliance meets innovation and growth knows no bounds.
Why Choose eLeaP?
Understand the compelling reasons and unique advantages for selecting eLeaP as your learning solution.
We know you have choices when it comes to learning management systems or people success platforms. Here’s why hundreds of companies have selected eLeaP. Here’s a hint, it is because of the value they derive each and every day from the system and service.
-
Brand
For over 19 years, we’ve been sound financial stewards of our brand, plowing back resources into our platforms so that our customers can continue to benefit from our stability.
-
Innovation
eLeaP uses a simple, minimalist but sophisticated philosophy which does not sacrifice innovation or breath for complexity and confusion.
-
Trust/Reliability
We ensure that the platform is tested and validated in our live development instance before integration to our production services.
-
Platform
Having one platform for managing online, in-person, instructor-led, e-commerce and extended enterprise learning and training is simply awesome.
-
Extended Services
We know that many of you come for the learning management system but end up staying for the extensive services we offer.
Case studies: a track record of excellence
See the results of so many who have used the eLeaP system
-

Bionical Solutions
Founded in 2001, Bionical Solutions is a pharmaceutical manufacturing company headquartered in Willington, Derbyshire, United Kingdom. The company provides clinical, commercial, and digital services to life sciences companies in the...
-

Biorasi, LLC.
Headquartered in Florida, United States, with office-based teams around the globe, Biorasi is a global, award-winning and full-service contract research organization (CRO) that delivers the speed and agility that today’s...
-

Natus Medical Incorporated
Founded in 1989, Natus Medical (Natus) is a medical equipment manufacturer based in the US. The company offers medical equipment, software, supplies, and services for diagnosing, monitoring, and treating impairments...
-

DealerStrong
Continuing Education
DealerStrong is a national retail automotive training and consulting company that works with clients to maximize their efforts and profits in the areas of Special Finance, Accounting, HR and BDC....